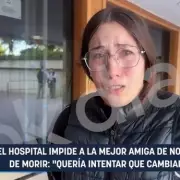
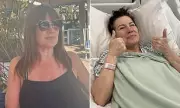
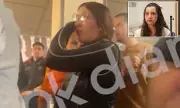Supplement Makers Push FDA to Allow Peptides and New Ingredients
Dietary supplement companies are urging the FDA to broaden ingredient criteria, potentially enabling peptides and probiotics in products, amid industry pressure and regulatory uncertainty.