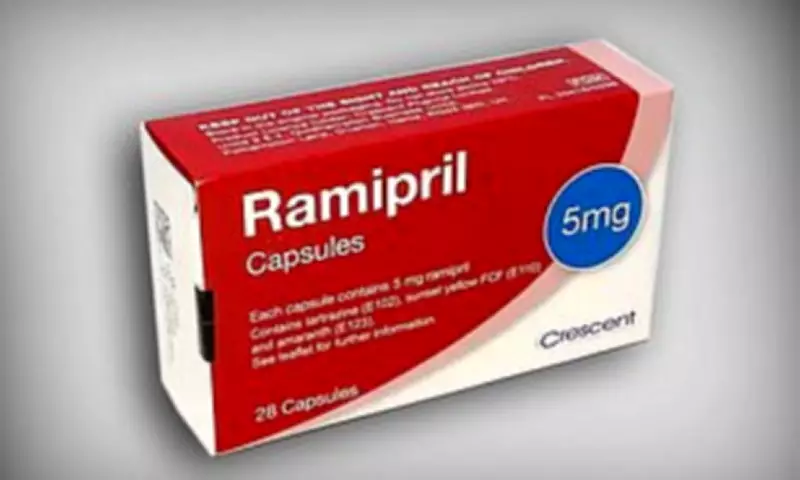
Patients taking a widely prescribed blood pressure medication are being issued an urgent safety alert after regulators discovered a packaging error that could see them receiving the wrong pills. The Medicines and Healthcare products Regulatory Agency (MHRA) has warned that certain packs of ramipril may have been accidentally filled with amlodipine tablets, which is another type of blood pressure drug that functions through a different biological mechanism.
Specific Batch and Manufacturer Identified
The alert specifically concerns 5mg ramipril packs manufactured by Crescent Pharma Limited. Individuals who are currently using this medication should immediately inspect their outer carton for the batch number GR164099, which is printed clearly on the packaging. If the blister strips inside are labelled as amlodipine rather than ramipril, the entire pack must be returned to the pharmacy without delay.
No Action Required for Correctly Labelled Packs
Importantly, packs that contain correctly labelled ramipril tablets do not need to be returned and can continue to be used as prescribed. The MHRA has clarified that the mix-up appears to have occurred during the packaging process at the factory, which produces both of these common cardiovascular medicines.
Low Risk but Potential for Dizziness
Because both ramipril and amlodipine are used to treat hypertension, the overall risk from this error is considered relatively low. However, the most probable side effect if the wrong tablets are consumed is dizziness, resulting from blood pressure dropping too sharply. The regulator has emphasised that anyone who suspects they have taken amlodipine by mistake and feels unwell should seek prompt medical advice, taking the medication and its leaflet with them to their GP or pharmacy.
Instructions for Pharmacies and Professionals
Pharmacies and healthcare professionals have also been directed to return any remaining stock from the affected batch. Shareen Doak, deputy director of benefit–risk evaluation at the MHRA, stated: 'If you have an affected pack and think you may have taken Amlodipine by mistake and are experiencing side effects, please seek immediate medical advice. Take the leaflet and any remaining tablets with you to your pharmacy or GP practice.' Any suspected adverse reactions should be reported via the MHRA Yellow Card scheme.
How These Medications Work Differently
Ramipril is classified as an ACE inhibitor, which lowers blood pressure by blocking a hormone that typically causes blood vessels to constrict. This action allows the vessels to relax and widen, reducing cardiac strain and enhancing blood flow throughout the body.
In contrast, amlodipine is a calcium channel blocker. It operates by preventing calcium from entering the muscle cells within blood vessel walls, thereby helping them to relax and dilate. This facilitates easier blood flow and a subsequent reduction in blood pressure.
Widespread Prescription and Usage
Both drugs are frequently prescribed to manage high blood pressure and diminish the risk of heart attacks and strokes, despite their distinct modes of action. Ramipril is available on prescription as tablets, capsules, or liquid and ranks among the UK's most commonly prescribed medicines, with over 35 million prescriptions issued between 2024 and 2025 alone.









