Unethical Vaccine Trial in Guinea-Bissau Sparks Global Alarm as Prototype for US Studies
Health experts are raising urgent concerns that an "unethical" vaccine trial conducted in Guinea-Bissau could serve as a "prototype" for future studies under the administration of Robert F Kennedy Jr, the US Secretary of Health and Human Services and a longtime vaccine critic. The trial, which focused on hepatitis B vaccines, has drawn scrutiny for its ethical lapses and potential influence on US public health policy.
Danish Researchers at the Heart of Controversial US Vaccine Policy
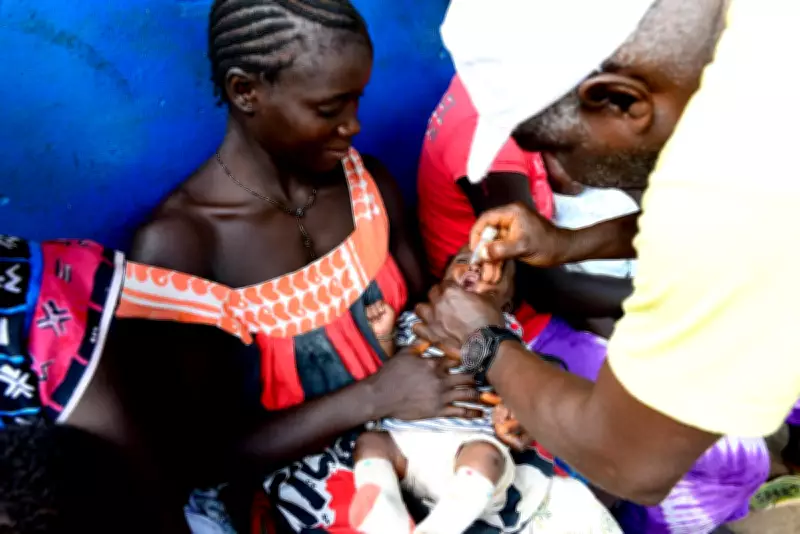
At the center of this controversy are Danish researchers from the Bandim Health Project, who have operated in Guinea-Bissau for 48 years. Their work on vaccine effects has been questioned, particularly a now-suspended trial that planned to vaccinate only half of newborns at birth, despite an 18% prevalence rate of hepatitis B in adults—a disease that can lead to severe and fatal health outcomes. This approach has been criticized as scientifically unsound and ethically disturbing.
Stand Up for Science, a US-based nonprofit, conducted an investigation in Guinea-Bissau, reviewing public records and interviewing experts. Their unreleased report, obtained by the Guardian, highlights deep entanglements between the Bandim Health Project and Guinea-Bissau's public health system, posing challenges for ethical research. Colette Delawalla, founder of Stand Up for Science, expressed fear that this trial could set a precedent for similar studies in the US, potentially leading to deadly consequences akin to historical ethical failures like the Tuskegee experiment.
Deep Ties to US Administration and Ethical Concerns
The Danish researchers have significant connections to the current US administration. RFK Jr has cited Peter Aaby, one of the researchers, as influential in shaping his views on vaccines. Christine Stabell Benn, another researcher, served on a hepatitis B working group for a US immunization advisory committee and hosted a podcast with Tracy Beth Høeg, a top FDA official who advocates for reducing "unnecessary" vaccines.
In Guinea-Bissau, the Bandim Health Project is described as being "embedded in the system," with the local ethics committee viewed as a "friends club" that charges fees for study reviews—a practice criticized as potentially biasing approvals. Magda Robalo, former health minister of Guinea-Bissau, noted that informed consent is extremely difficult due to low literacy rates and language barriers, such as the local term for hepatitis B being confused with "yellow fever."
Robalo emphasized that Guinea-Bissau lacks credible public health research institutions, leading many local researchers to collaborate with Bandim, creating conflicts of interest. For instance, Armando Sifna, the current public health director, has been affiliated with Bandim while holding government positions, raising questions about impartiality.
Global Responses and Calls for Action
Following public outcry, the University of Southern Denmark has paused all work on the trial pending an independent ethical review by the WHO. The US House Committee on Energy and Commerce has labeled the trial "ethically disturbing and scientifically unsound," questioning the CDC's role in funding it. WHO Director-General Tedros Adhanom Ghebreyesus condemned the study as unethical, citing concerns over scientific justification and ethical safeguards.
Guinea-Bissau's foreign minister has stated the trial will not proceed, and African health officials, including from the Africa CDC, are prioritizing the rollout of hepatitis B vaccines for all newborns by 2027. The US allocated $1.6 million to the trial, with matching funds from foundations, leading critics to argue that this money could instead vaccinate every newborn in Guinea-Bissau for years.
This case has resonated across Africa, prompting efforts to strengthen local research capabilities and reduce dependency on Global North institutions. As the review continues, the implications for US health policy under RFK Jr remain a critical concern for global public health.









