The U.S. Food and Drug Administration (FDA) granted approval on Wednesday to a groundbreaking oral treatment for psoriasis, a development that could bring significant relief to millions of individuals grappling with this chronic skin condition. The agency has officially sanctioned Johnson & Johnson's novel pill, Icotyde, for use in patients aged 12 years and older who are diagnosed with moderate to severe plaque psoriasis.
A Needle-Free Alternative for Psoriasis Management
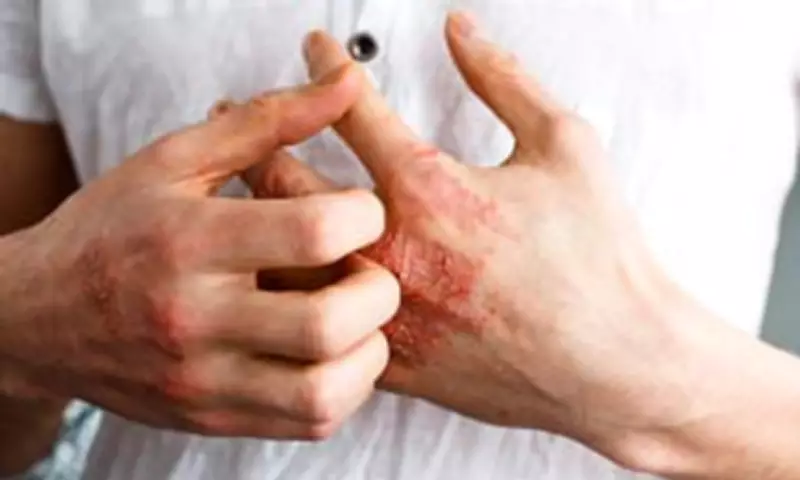
This innovative oral medication is engineered to replicate the therapeutic effects of existing psoriasis treatments but eliminates the necessity for invasive needles and injections. Psoriasis is an autoimmune disorder characterized by the rapid accumulation of skin cells, resulting in the formation of itchy, flaky, scaly, and inflamed skin patches known as plaques. The condition is influenced by genetic factors and can be triggered by environmental elements such as stress or infections.
Although psoriasis is not typically life-threatening, it often induces profoundly uncomfortable symptoms. These include persistent itching and burning sensations, dry and cracked skin that may bleed, and swollen, painful joints. Beyond the physical discomfort, many sufferers experience considerable anxiety and embarrassment related to their condition, leading to substantial mental health distress alongside the physical manifestations.
Current Treatment Landscape and Limitations
Present treatment options encompass topical steroid creams, phototherapy using artificial light to decelerate skin cell growth, and injectable biologics. These alternatives can incur annual costs reaching thousands of dollars, even with insurance coverage, and are generally less convenient compared to a simple daily pill regimen.
Icotyde represents a pioneering advancement as the first and only oral therapy for psoriasis that specifically targets the Interleukin-23 (IL-23) receptor. This protein complex resides on immune cells and binds to IL-23 cytokines, which are molecules that provoke inflammation. While inflammation is a crucial component of the body's infection-fighting response, excessively active levels can precipitate autoimmune diseases like psoriasis.
Clinical Trials and Safety Profile
The FDA's approval is underpinned by encouraging outcomes from four Phase 3 clinical trials, which involved over 2,500 participants. Research indicates that Icotyde boasts a favorable safety profile, with minor side effects reported. These adverse effects include headaches, nausea, cough, fungal infections, and fatigue. However, the safety of the drug for pregnant or breastfeeding women remains uncertain and requires further investigation.
Pricing and Insurance Considerations
Johnson & Johnson has yet to disclose the list price for Icotyde. For context, injectable medications that target IL-23 can command prices as high as $100,000 per year. It is also currently unclear whether insurance providers will extend coverage for this new oral treatment, which could impact its accessibility for many patients.
Expert Commentary on Icotyde's Potential
Dr. Linda Stein Gold, the Director of Dermatology Clinical Research at Henry Ford Health, remarked, 'Icotyde delivers something unique in psoriasis treatment – combining skin clearance with a favorable safety profile in a once-daily pill, making it an easy addition to a patient’s routine.'
She further elaborated, 'With new guidance from the International Psoriasis Council that clarifies when to move beyond cycling on topical treatments to systemic therapy, an innovative option like Icotyde is a potential game-changer for many adult and adolescent patients.' This endorsement underscores the transformative potential of Icotyde in enhancing the quality of life for those affected by psoriasis, offering a more convenient and effective therapeutic pathway.









